The burned hand teaches best. After that, advice about fire goes to the heart.
J. R. R. Tolkein, The Two Towers (1954)
As is often the case in an educational context, and with all due respect to Tolkein, I think Siegfried Engelman actually said it best.
The physical environment provides continuous and usually unambiguous feedback to the learner who is trying to learn physical operations . . .
Siegfried Engelmann and Douglas Carnine, Theory of Instruction (1982)
I am going to outline a practical approach that will help students understand that black objects are good emitters and good absorbers of infrared radiation.
What I propose is a simple, inexpensive and low risk procedure (similar to this one from the IoP) that won’t actually inflict any actual “burned hands” but will, hopefully, through a clever (imho) manipulation of the physical environment, speak directly to the heart — or at least to students’ “sense of mechanism” about how the world works.
Half human and half infrared detector
Obtain tubes of matt black and white facepaint. (These are typically £5 or less.) Choose a brand that is water based for easy removal and is compliant with EU and UK regulations.
We also need a good source of infrared radiation. Some suppliers such as Nicholl and Timstar can supply a radiant heat source that is safe to use in schools. Although these can be expensive to purchase, there may already be one hiding in a cupboard in your school. If you don’t have one, use a 60W filament light bulb mounted in desk lamp (do not use a fluorescent or LED lamp — they don’t produce enough IR!). Failing that, you could use a raybox with a 24W, 12V filament lamp to act as the infrared source. [UPDATE: Paul Bushen also recommends a more economical option — an infrared heat lamp.)
Use the facepaint to make 2 cm by 2 cm squares on the back of one hand in black and in white on the other. Hold each square up to the infrared source so they are a similar distance from it.
Hold the hands still in front of the source for a set time. This could be anywhere between five seconds and a few tens of seconds, depending on the intensity of the source. You should run through this experiment ahead of time to make sure that there is minimal risk of any serious burns for the time you intend to allocate. If you are using rayboxes then you might need a separate one for each hand.
The hand with the black paint becomes noticeably warmer when exposed to infrared radiation. We can deduce that this is because the colour black is better and absorbing the infrared than the white colour.
Energy is being transferred via light into the thermal energy store of the hand.
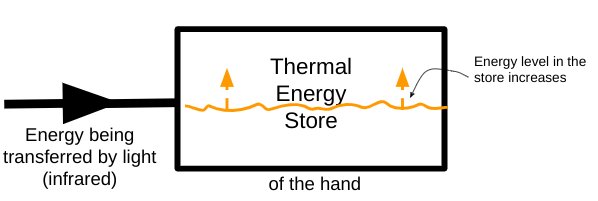
We can use a black painted hand as a rudimentary detector for infrared. The hotter it gets, the more infrared is being emitted.
Enter Leslie’s cube . . .

Fill a Leslie’s cube with hot water from a kettle and then get students to place the hand with the black square a couple of centimetres away from the black face of the cube. After a few seconds, ask them to place the same hand by the white face of the cube. (Although, for the best contrast, you should maybe try the polished silver side). Make sure the student’s hand does not actually touch the face of the Leslie’s cube, otherwise they may end up with an actual burned hand!
The fact that the black face emits more infrared radiation is immediately directly perceivable by the “infrared detector” hand which feels distinctly warmer than when it’s placed next to the black coloured face rather than the white face.
This procedure is, I think, more convincing to many students as opposed to merely using (say) a digital infrared detector and reading off a larger number from the dark side compared to the white side.
So why aren’t more domestic CH radiators painted black, rather than white ?
via quora (other answers available):
Richard A. Nichols III
Updated 1 year ago ·
Black absorbs light/heat energy, white reflects it. So if you want to keep as much heat outside the radiator as you can (and the point is to move heat from the inside of the radiator to outside of it), then you’d paint it white.
Maybe Santiago is right and the physics difference is negligible, and maybe it’s just an aesthetic decision, I don’t know, but if you cared about which color to make the radiator for physics reasons, I think it’d be white.
Someone else’s edit: Actually this is wrong: black absorbs and emits heat better than all other colours, so theoretically black would be most effective at absorbing heat from inside the radiator and emitting it outside. White and silver are best at reflecting heat (back into radiator / back to room) so this is of little use if you want to heat a cooler room. This is. Why I’m trying to work out why white radiators are deemed most effective.
My thought is that they aren’t actually radiators — they’re convectors because they are designed to heat the space by convection. In this case, you’d want the thermal energy transferred by conduction to the air next to the “radiator” and then that air would rise and form a convection current that would, over time, gradually heat up all the air in the room. To maximise that initial energy transfer by conduction, you want to minimise the energy transfer by radiation — hence, so-called “radiators” are painted white. This also explains why you can place soft furnishings in front of “radiators” without losing the heating effect of the “radiators”.