The AQA GCSE Science specification calls for students to understand and apply the concepts of not only thermal energy stores but also internal energy. What follows is my understanding of the distinction between the two, which I hope will be of use to all science teachers.
My own understanding of this topic has undergone some changes thanks to some fascinating (and ongoing) discussions via EduTwitter.
What I suggest is that we look at the phenomena in question through two lenses:
- a macroscopic lens, where we focus on things we can sense and measure directly in the laboratory
- a microscopic lens, where we focus on using the particle model to explain phase changes such as melting and freezing.
Thermal Energy Through the Macroscopic Lens
 The enojis for thermal energy stores (as suggested by the Institute of Physics) look like this (Note: ‘enoji’ = ‘energy’ + ’emoji’; and that the IoP do not use the term):
The enojis for thermal energy stores (as suggested by the Institute of Physics) look like this (Note: ‘enoji’ = ‘energy’ + ’emoji’; and that the IoP do not use the term):
In many ways, they are an excellent representation. Firstly, energy is represented as a “quasi-material entity” in the form of an orange liquid which can be shifted between stores, so the enoji on the left could represent an aluminium block before it is heated, and the one on the right after it is heated. Secondly, it also attempts to make clear that the so-called forms of energy are labels added for human convenience and that energy is the same basic “stuff” whether it is in the thermal energy store or the kinetic energy store. Thirdly, it makes the link between kinetic theory and thermal energy stores explicit: the particles in a hot object are moving faster than the particles in the colder object.
However, I think the third point is not necessarily an advantage as I believe it will muddy the conceptual waters when it comes to talking about internal energy later on.
If I was a graphic designer working for the IoP these are the enojis I would present: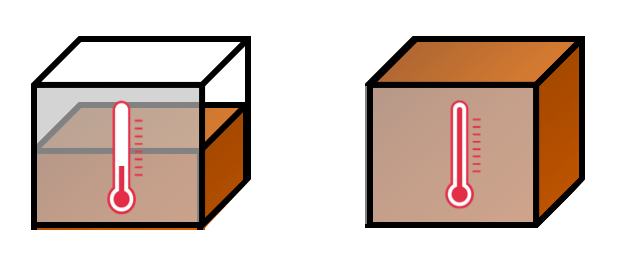
In other words, a change in the thermal energy store is always associated with a temperature change. To increase the temperature of an object, we need to shift energy into the thermal energy store. To cool an object, energy needs to be shifted out of the thermal energy store.
This has the advantage of focusing on the directly observable macroscopic properties of the system and is, I think, broadly in line with the approach suggested by the AQA specification.
Internal Energy Through the Microscopic Lens
Internal energy is the “hidden” energy of an object.
The “visible” energies associated with an object would include its kinetic energy store if it is moving, and its gravitational potential energy store if it is lifted above ground level. But there is also a deeper, macroscopically-invisible store of energy associated with the particles of which the object is composed.
To understand internal energy, we have to look through our microscopic lens.
The Oxford Dictionary of Physics (2015) defines internal energy as:
The total of the kinetic energies of the atoms and molecules of which a system consists and the potential energies associated with their mutual interactions. It does not include the kinetic and potential energies of the system as a whole nor their nuclear energies or other intra-atomic energies.
In other words, we can equate the internal energy to the sum of the kinetic energy of each individual particle added to the sum of the potential energy due to the forces between each particle. In the simple model below, the intermolecular forces between each particle are modelled as springs, so the potential energy can be thought as stretching and squashing the “springs”. (Note: try not to talk about “bonds” in this context as it annoys the hell out of chemists, some of whom have been known to kick like a mule when provoked!)
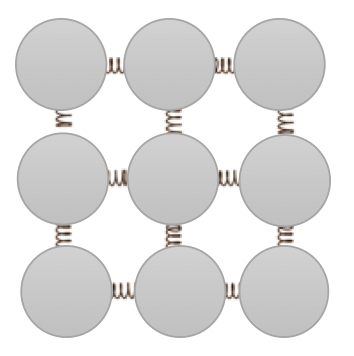
We can never measure or calculate the value of the absolute internal energy of a system in a particular state since energy will be shifting from kinetic energy stores to potential energy stores and vice versa moment-by-moment. What is a useful and significant quantity is the change in the internal energy, particularly when we are considering phase changes such as solid to liquid and so on.
This means that internal energy is not synonymous with thermal energy; rather, the thermal energy of a system can be taken as being a part (but not the whole) of the internal energy of the system.
As Rod Nave (2000) points out in his excellent web resource Hyperphysics, what we think of as the thermal energy store of a system (i.e. the sum of the translational kinetic energies of small point-like particles), is often an extremely small part of the total internal energy of the system.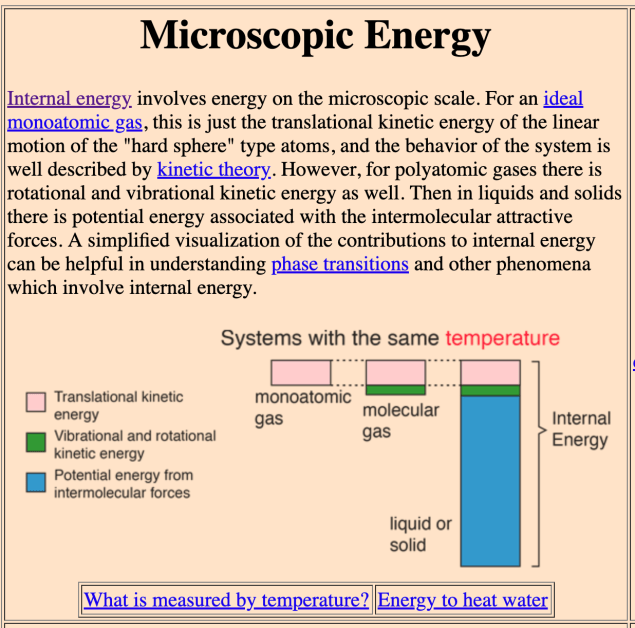
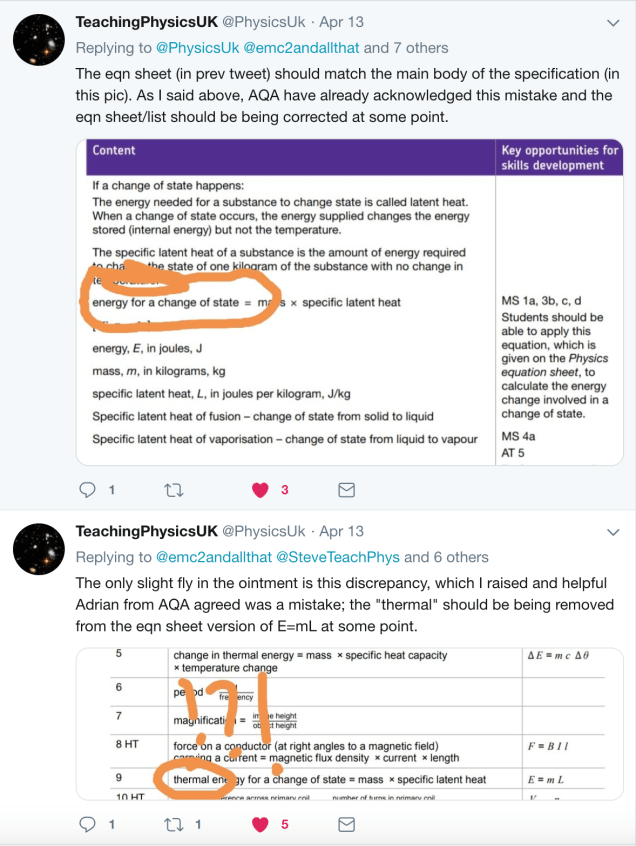
AQA: Oops-a-doodle!
My excellent Edu-tweeting colleague @PhysicsUK has pointed out that there is indeed a discrepancy between the equations presented by AQA in their specification and on the student equation sheet.
If a change in thermal energy is always associated with a change in temperature (macroscopic lens) then we should not use the term to describe the energy change associated with a change of state when there is no temperature change (microscopic lens).
@PhysicsUK reports that AQA have ‘fessed up to the mistake and intend to correct it in the near future. Sooner would be better than later, please, AQA!
References
Nave, R. (2000). HyperPhysics. Georgia State University, Department of Physics and Astronomy.

Reblogged this on The Echo Chamber.