“Something with a lot of energy will kill you.”
This has stayed with me from my PGCE course at Swansea University, many years ago. It was said by Frank Banks, the course tutor, in response to the question “What’s the simplest way to describe energy?”
And as pithy descriptions of energy go, it’s not half-bad. A small stone, dropped from the top of a skyscraper: lots of energy before it hits the ground — it could kill you. A grand piano, dropped from six feet above your head: lots of energy — it could kill you. Licking your fingers and touching the bare live and neutral wires in a socket: the conduction electrons in your body suddenly acquire a lot of energy — and yes, they could kill you. (With alternating current, of course, the electrons that will kill you are already inside your body — freaky!)
This attention-grabbing definition of energy seems to lead naturally to a more formal definition of “Energy is the capacity to do work“. This still leaves the problem of defining work, of course, but as R. A. Lafferty once said, that’s another and much more unpleasant story.
As I mentioned in an earlier post, I have been writing the Energy scheme of work for GCSE Science. As part of that brief, I wrote a short summary for my science colleagues of the IoP’s new approach to energy. I present it below without much amendment (or even a proper spellcheck) in the hope that someone, somewhere, at some time — may find it useful 🙂
The problem with teaching energy
One reason for the difficulty in deciding what to say about energy at school level is that the scientific idea of energy is very abstract. It is, for example, impossible to say in simple language what energy is, or means. Another problem is that the word ‘energy’ has entered everyday discourse, with a meaning that is related to, but very different from, the scientific one. [ . . .]
This ‘forms of energy’ approach has, however, been the subject of much debate. One criticism is that pupils just learn a set of labels, which adds little to their understanding. For example, one current textbook uses the example of a battery powered golf buggy. It asks pupils to think of this in the following terms:
Chemical energy in the battery is transformed into electrical energy which is carried by the wires to the motor. The motor then transforms this into kinetic energy as the buggy moves.
This, however, adds nothing to the following explanation, which does not use energy ideas:
The battery supplies an electric current which makes the motor turn. This then makes the buggy move.
A good general rule when explaining anything is that you should use the smallest number of ideas needed to provide an explanation, and not introduce any that are unnecessary
Robin Millar [2012] http://www.lancsngfl.ac.uk/nationalstrategy/ks3/science/files/TeachingAboutEnergy(RobinMillar).pdf
Energy – a new hope (!)
The new approach to the teaching of energy developed by the Institute of Physics (IoP) suggests that we limit our consideration of energy to situations where we might want to do calculations (at KS4, KS5 or beyond).
We should talk of energy being stored and shifted. The emphasis should be on the start and end of the process with minimal attention being given to any intermediate stages.
Consider the following examples:
- lifting an object. Chemical potential energy store is emptied, and gravitational potential energy store is filled (note that we are not interested in intermediate motion as it doesn’t affect the final energy store).
- rolling an object down a slope to the bottom. Gravitational potential energy store is emptied and thermal energy stores (of slope, of pen) increased.
- Boiling water in kettle. Chemical store (from coal/gas power station) is emptied. Thermal store of water increased, thermal store of air increased, thermal store of kettle increased.
[Examples taken from http://scientistshavesaid.blogspot.co.uk/2013/02/teaching-energy.html]
The new approach has been adopted by all UK exam boards for their new specs and is used in the AQA approved textbooks.
The following energy stores are considered: kinetic energy store, gravitational potential energy store, elastic potential energy store, thermal energy store, chemical potential energy store, nuclear energy store, vibrational energy store, electromagnetic energy store (note: the last is limited to situations involving static electric charges and static magnetic poles in magnetic fields).[NB Items in bold are those required for GCSE Combined Science.]
One major difference is that electric current and light are no longer considered as forms of energy. Rather, these are now regarded as means of transferring energy.
Rise of the Enojis
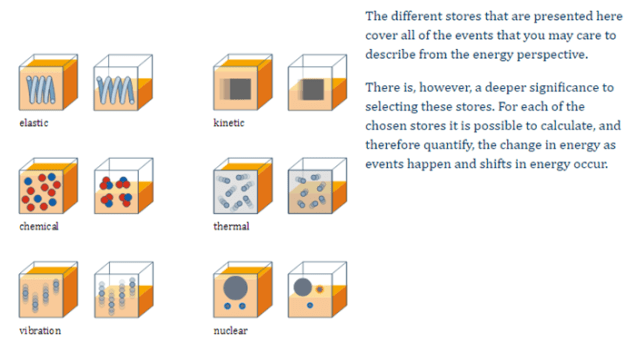
[Image from http://www.supportingphysicsteaching.net/En02PN.html]
I suggest these energy icons should be called enojis (by analogy with emojis).
Probably the biggest adjustment for most teachers will be to avoid referring to light and sound as forms of energy and to treat them as pathways for transferring energy instead.
“Energy is the new orange” and summary
More (much more!) on the IoP’s “energy as an orange liquid” model can be found at http://www.supportingphysicsteaching.net/En02TL.html and http://www.supportingphysicsteaching.net/En02PN.html.


Reblogged this on The Echo Chamber.
“Probably the biggest adjustment for most teachers will be to avoid referring to light and sound as forms of energy” Hmmm especially after such seemed a big feature of the first (explosive) RI Christmas lecture with emphasis on the sun/earthly fusion/… all strongly represented photon wise. Why is E=hf screaming in my head.
It makes me go Hmmm as well, and I think I will find it hard to adjust. But then, perhaps the advice should read “… referring to light and sound as ENERGY STORES.”
Energy is energy, after all. So photons carry energy, but is it helpful to think of light as an energy STORE which can be modelled as a container of orange liquid…? A single photon cannot be partially absorbed after all, and it’s energy is unavailable until it has been absorbed. The one example I can think of where it might be helpful to think of light as an energy store is the Radiative Zone of a star, perhaps…?
“a container of orange liquid” should go down well in these parts
I would never say no to a chilled container of orange liquid (shaken, not stirred).
And that is where we find ourselves confused, as physics teachers trying to deal with the new terminology and methods…..there is no ‘light energy store’ but instead energy is only transferred by EM (light) radiation…or something.
Very cumbersome indeed when trying to explain something as simple as “light energy from the sun is converted into chemical energy in a plant by photosynthesis” in terms of energy stores. Thankfully GCSE chemistry and biology specifications make no mention of energy stores.
Thanks for the comment! They are always really appreciated.
Over the last few years, I’ve gradually come to the conclusion that the IoP is right to discourage the use of ‘light energy’. My thinking (and you may well disagree) is that it doesn’t exist in a ‘locate-able’ space. The GCSE Biology specs are written to be consistent with the Energy Stores and Pathways model, so they talk about ‘energy transferred by light’ rather than light energy. Since Energy Stores and Pathways is mainly concerned with macroscopic phenomena it doesn’t feature heavily in the GCSE Chem spec since that focuses on microscopic energy changes. However, the language seems carefully neutral so as not tread of the IoP’s toes(!)
Photosynthesis *can* (imo) be explained simply using the Stores and Pathways model: https://physicsteacher.blog/2021/01/01/photosynthesis-and-energy-stores/
Many thanks for taking the time to comment, and feel free to agree/disagree/ignore 🙂
Now get their minds round potential energy.
Elastic, gravitational or chemical? (It’s like the bridge scene from Monty Python And The Holy Grail: “African or European sparrow?” https://youtu.be/y2R3FvS4xr4
“rolling an object down a slope to the bottom. Gravitational potential energy store is emptied and thermal energy stores (of slope, of pen) increased.”
Has the brick lost ALL its potential energy at the bottom of the slope?
Have we forgotten all about negative numbers?
I think you make an interesting point that highlights the difficulty of talking correctly about energy even at a simple level. Of course, gpe is a moveable feast as far as assigning zero: we can place the zero where we wish for our own convenience — but would any of us want to introduce gravitational potential at ks3 or 4? That said, the IoP implies that although the GP store has had energy removed, it hasn’t necessarily completely emptied.
I’m not 100% confident with the IoP model yet, but it does seem to have the scope to regularise and standardise the way we talk about energy at a school level, which had become a dog’s dinner of confusing interpretations.
I am tempted to suggest that the word “store” be enclosed in brackets (parentheses).