Nuclear binding energy and binding energy per nucleon are difficult concepts for A-level physics students to grasp. I have found the ‘pool table analogy’ that follows helpful for students to wrap their heads around these concepts.
Background
Since mass and energy are not independent entities, their separate conservation principles are properly a single one — the principle of conservation of mass-energy. Mass can be created or destroyed , but when this happens, an equivalent amount of energy simultaneously vanishes or comes into being, and vice versa. Mass and energy are different aspects of the same thing.
Beiser 1987: 29
E = mc2
There, I’ve said it. This is the first time I have directly referred to this equation since starting this blog in 2013. I suppose I have been more concerned with the ‘andallthat‘-ness side of things rather than E=mc2. Well, no more! E=mc2 will form the very centre of this post. (And about time too!)
The E is for ‘rest energy’: that is to say, the energy an object of mass m has simply by virtue of being. It is half the energy that would be liberated if it met its antimatter doppelganger and particles and antiparticles annihilated each other. A scientist in a popular novel sternly advised a person witnessing an annihilation event to ‘Shield your eyes!’ because of the flash of electromagnetic radiation that would be produced.
Well, you could if you wanted to, but it wouldn’t do much good since the radiation would be in the form of gamma rays which are to human eyes what the sound waves from a silent dog whistle are to human ears: beyond the frequency range that we can detect.
The main problem is likely to be the amount of energy released since the conversion factor is c2: that is to say, the velocity of light squared. For perspective, it is estimated that the atomic bomb detonated over Hiroshima achieved its devastation by directly converting only 0.0007 kg of matter into energy. (That would be 0.002% of the 38.5 kg of enriched uranium in the bomb.)
Matter contains a lot of energy locked away as ‘rest energy’. But these processes which liberate rest energy are mercifully rare, aren’t they?
No, they’re not. As Arthur Beiser put it in his classic Concepts of Modern Physics:
In fact, processes in which rest energy is liberated are very familiar. It is simply that we do not usually think of them in such terms. In every chemical reaction that evolves energy, a certain amount of matter disappears, but the lost mass is so small a fraction of the total mass of the reacting substances that it is imperceptible. Hence the ‘law’ of conservation of mass in chemistry.
Beiser 1987: 29
Building a helium atom
The constituents of a helium nucleus have a greater mass when separated than they do when they’re joined together.
Here, I’ll prove it to you:

The change in mass due to the loss of energy as the constituents come together is appreciable as a significant fraction of its original mass. Although 0.0293/4.0319*100% = 0.7% may not seem like a lot, it’s enough of a difference to keep the Sun shining.
The loss of energy is called the binding energy and for a helium atom it corresponds to a release of 27 MeV (mega electron volts) or 4.4 x 10-12 joules. Since there are four nucleons (particles that make up a nucleus) then the binding energy per nucleon (which is a guide to the stability of the new nucleus) is some 7 MeV.
But why must systems lose energy in order to become more stable?
The Pool Table Analogy for binding energy
Imagine four balls on a pool table as shown.
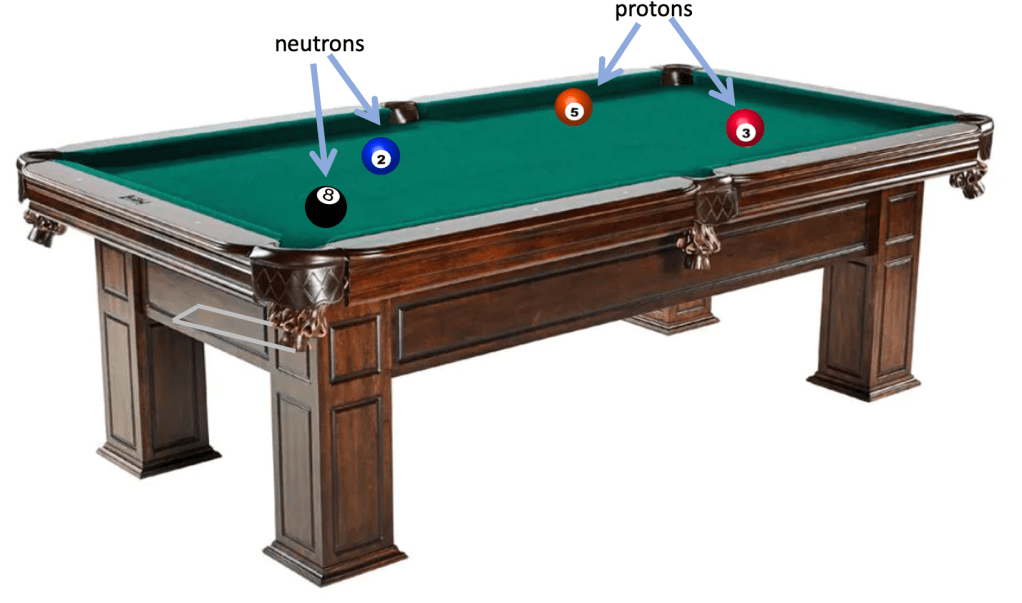
The balls have the freedom to move anywhere on the table in their ‘unbound’ configuration.
However, what if they were knocked into the corner pocket?

To enter the ‘bound’ configuration they must lose energy: in the case of the pool balls we are talking about gravitational potential energy, a matter of some 0.30 J per ball or a total energy loss of 4 x 0.30 = 1.2 joules.
The binding energy of a pool table ‘helium nucleus’ is thus some 1.2 joules while the ‘binding energy per nucleon’ is 0.30 J. In other words, we would have to supply 1.2 J of energy to the ‘helium nucleus’ to break the forces binding the particles together so they can move freely apart from each other.
Just as a real helium nucleus, the pool table system becomes more stable when some of its constituents lose energy and less stable when they gain energy.
Reference
Beiser, A. (1987). Concepts of modern physics. McGraw-Hill Companies.